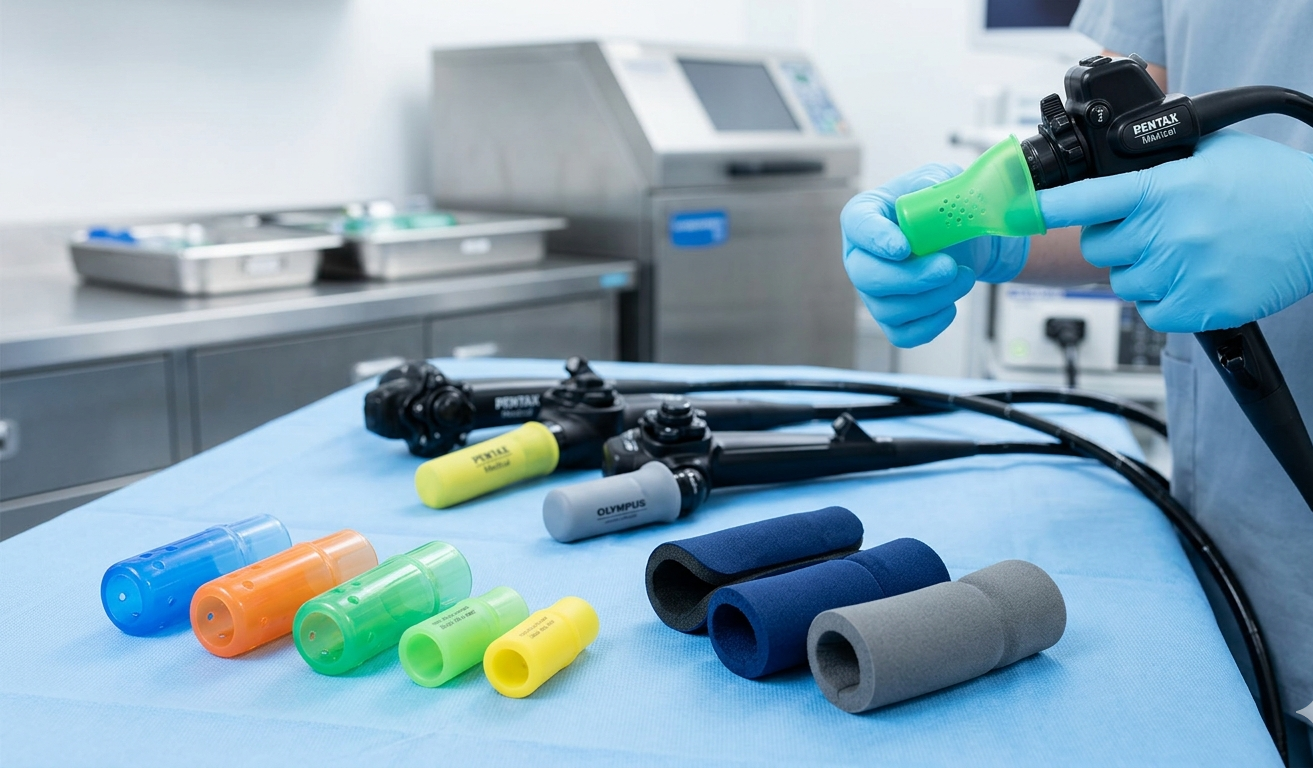
Endoscope tip protectors are specialized medical accessories designed to safeguard the sensitive distal tips of endoscopes from physical damage during transportation, storage, and handling between procedures. As the global healthcare infrastructure expands and endoscopic procedures become more ubiquitous across multiple medical specialties, the market for endoscope tip protectors is witnessing sustained growth. The evolution of endoscopy equipment, paired with rigorous reprocessing protocols, has made protective accessories a critical component of healthcare quality management.
Endoscope Tip Protectors Market Scope
The Endoscope Tip Protectors Market Scope encompasses a wide array of product types, end-use applications, materials, and geographic regions. From a product standpoint, the market includes single-use disposable tip protectors, reusable tip protectors, and specialty variants designed for niche endoscope types. End-user segments span hospitals, ambulatory surgical centers, specialty clinics, and diagnostic imaging facilities. The scope of the market also extends across gastrointestinal endoscopy, bronchoscopy, cystoscopy, and arthroscopy applications, reflecting the diverse use cases of endoscopic technology. Geographically, the scope covers North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa, each region presenting unique growth opportunities driven by distinct healthcare dynamics.
Get Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00013457
Product Segmentation and Applications
Within the market's broad scope, product segmentation plays a pivotal role in defining competitive positioning. Silicone-based tip protectors dominate due to their flexibility, chemical resistance, and compatibility with endoscope disinfection chemicals. Polyurethane and foam-based variants are gaining traction for their cost-effectiveness and cushioning properties. Gastrointestinal endoscopy remains the largest application segment, reflecting the high volume of colonoscopies and upper GI scopes performed globally.
Regulatory Framework
The regulatory scope governing endoscope tip protectors is substantial. In the United States, these products are regulated as Class I or Class II medical devices by the FDA, depending on their design and indications. The European Union's Medical Device Regulation (MDR) imposes rigorous conformity assessments for products entering the EU market. Compliance with ISO standards related to biocompatibility and sterilization further narrows the field to manufacturers with robust quality management systems.
Market Opportunities
The market scope continues to expand as healthcare facilities in emerging economies invest in upgrading their endoscopy infrastructure. Government health initiatives aimed at improving gastrointestinal and respiratory healthcare are creating new demand pockets in Southeast Asia, Africa, and Latin America. Additionally, the rising adoption of telemedicine and remote diagnostic platforms is opening discussions around portable endoscopy setups, indirectly driving demand for compact and lightweight tip protection solutions.
Related Report:
1) Examination Lights Market Share, Growth & Demand by 2034
2) Patient Recliners Market Trends, Demand & Growth by 2034
3) First Aid Kit Market Share, Size & Demand by 2034
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Information –
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean German Japanese French Chinese Italian Spanish