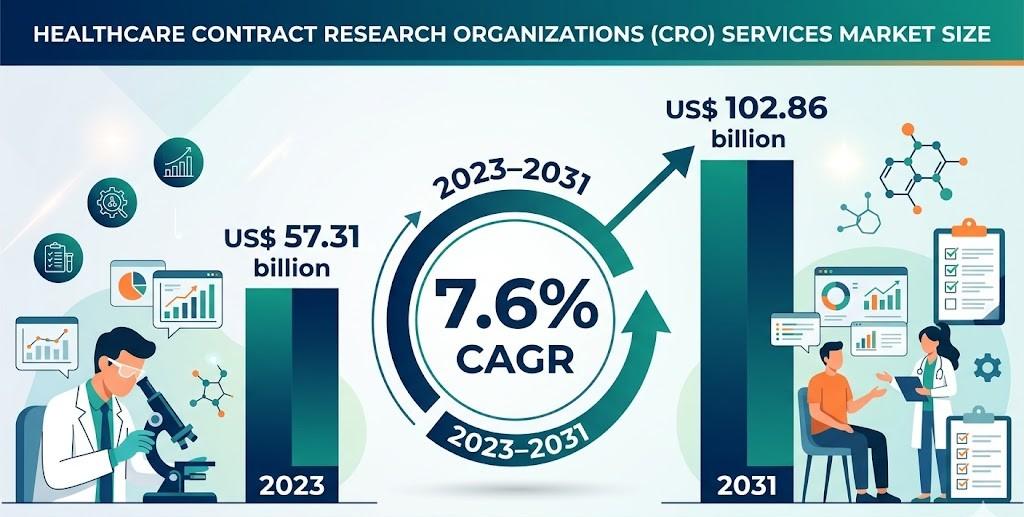
The global ecosystem for outsourced clinical development has become a cornerstone of modern healthcare innovation. Pharmaceutical, biotechnology, and medical device companies increasingly rely on external partners to accelerate research timelines, manage complex trials, and navigate regulatory requirements. This shift reflects the growing need for efficiency, flexibility, and specialized expertise in drug development processes.
The Healthcare Contract Research Organizations (CRO) Services Market Drivers are closely linked to the rapid increase in clinical research activities, rising disease burden, and expanding demand for innovative therapeutics. As organizations seek to optimize costs and improve success rates, CROs are emerging as strategic collaborators across the entire development lifecycle.
Increasing Clinical Trials and Disease Burden
One of the primary growth drivers is the significant rise in global clinical trials. The increasing prevalence of chronic diseases such as asthma, cardiovascular disorders, and neurological conditions has intensified the demand for new treatment options. With millions of patients requiring long-term care, the need for continuous drug development remains strong.
Clinical trials require substantial infrastructure, skilled personnel, and regulatory expertise. CROs provide end-to-end solutions, including study design, patient recruitment, data analysis, and compliance management. These capabilities allow sponsors to focus on innovation while outsourcing operational complexities. As the number of trials continues to grow, the reliance on CRO services is expected to expand further.
Rising Outsourcing Strategies Among Pharma and Biotech Firms
Outsourcing has become a key operational strategy for pharmaceutical and biotechnology companies. Instead of maintaining extensive in-house capabilities, organizations are partnering with CROs to access specialized knowledge and scalable resources. This approach helps reduce operational costs and enhances efficiency in managing clinical programs.
Strategic collaborations between CROs and sponsors are increasing, enabling faster execution of research projects. Partnerships focused on early phase development, biologics research, and clinical trial management are becoming more common. These alliances not only improve productivity but also contribute to higher success rates in drug approvals.
Additionally, restructuring initiatives within major companies have led to the creation of independent CRO entities, further strengthening the outsourcing ecosystem.
Growth in Biologics and Biosimilars Development
The expansion of biologics and biosimilars is another significant factor driving demand for CRO services. Advances in biotechnology, including genomics, proteomics, and biomarker research, are transforming the drug development landscape. These innovations require specialized expertise and sophisticated infrastructure, which CROs are well-positioned to provide.
Emerging markets, particularly in Asia Pacific, are witnessing increased investments in biotechnology. Government initiatives aimed at promoting innovation and high-tech manufacturing are supporting the development of advanced therapeutics. As a result, CROs are experiencing growing demand for services related to biologics and biosimilars research.
The increasing focus on personalized medicine and targeted therapies further amplifies the need for complex clinical trials, reinforcing the role of CROs in the healthcare ecosystem.
Expanding R&D Investments and Funding Support
Rising investments in research and development are playing a crucial role in driving the adoption of CRO services. Pharmaceutical and biotechnology companies are allocating substantial budgets to develop new drugs and therapies, particularly for chronic and rare diseases.
Government agencies and private organizations are also providing funding support for clinical research. These initiatives encourage innovation and facilitate the execution of large-scale trials. CROs benefit from this trend by offering cost-effective and efficient solutions that align with funding objectives.
Moreover, the increasing complexity of regulatory requirements necessitates expert guidance, further boosting the demand for CRO services. Organizations prefer outsourcing to ensure compliance with evolving global standards.
Download Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00010465
Regional Expansion and Emerging Opportunities
Geographically, North America continues to dominate due to strong R&D infrastructure and a high number of clinical trials. The presence of established pharmaceutical companies and stringent regulatory frameworks supports the growth of CRO services in the region.
Asia Pacific is emerging as a high-growth region, driven by cost advantages, skilled workforce availability, and supportive government policies. Countries such as China and India are becoming prominent hubs for clinical research and biotechnology development.
The expansion into emerging markets offers significant opportunities for CROs to diversify their service offerings and strengthen their global footprint. Increasing adoption of advanced technologies and digital tools is further enhancing operational efficiency and data management capabilities.
Key Players in the Healthcare Contract Research Organizations (CRO) Services Sector
- Charles River Laboratories Inc.
- IQVIA Inc.
- Laboratory Corporation of America Holdings
- Parexel International Corporation
- ICON Plc.
- PRA Health Sciences
- Syneos Health
- Medpace
- WuXi AppTec
- PPD Inc.
These players are focusing on expanding their service portfolios, leveraging advanced technologies, and forming strategic partnerships to strengthen their market position.
Future Outlook
The Healthcare Contract Research Organizations (CRO) Services Market Drivers are expected to remain strong over the coming years, supported by continuous advancements in medical research and increasing demand for innovative therapies. The integration of digital technologies such as artificial intelligence, decentralized trials, and real-world data analytics is likely to redefine clinical development processes.
Related Reports @
Healthcare e-Commerce Sodium Market Report 2034
Healthcare Barcode Printer Market Growth, Size, Share, Trends, Key Players Analysis, and Forecast till 2034
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish