Viral inactivation has become a critical component in ensuring the safety and efficacy of biopharmaceutical products by eliminating viral contaminants during manufacturing processes. These technologies are widely applied in the production of vaccines, blood derivatives, and biologics, where maintaining product purity is essential.
Increasing Demand for Biopharmaceutical Safety
The growing emphasis on product safety and regulatory compliance is significantly driving the Viral Inactivation Market. Biopharmaceutical manufacturers are increasingly adopting advanced viral inactivation techniques to meet stringent quality standards and ensure patient safety. The rising prevalence of chronic and infectious diseases has further accelerated the need for biologics, thereby increasing the demand for reliable viral safety solutions. Additionally, continuous investments in research and development are supporting the adoption of innovative inactivation technologies.
Viral Inactivation Market Growth
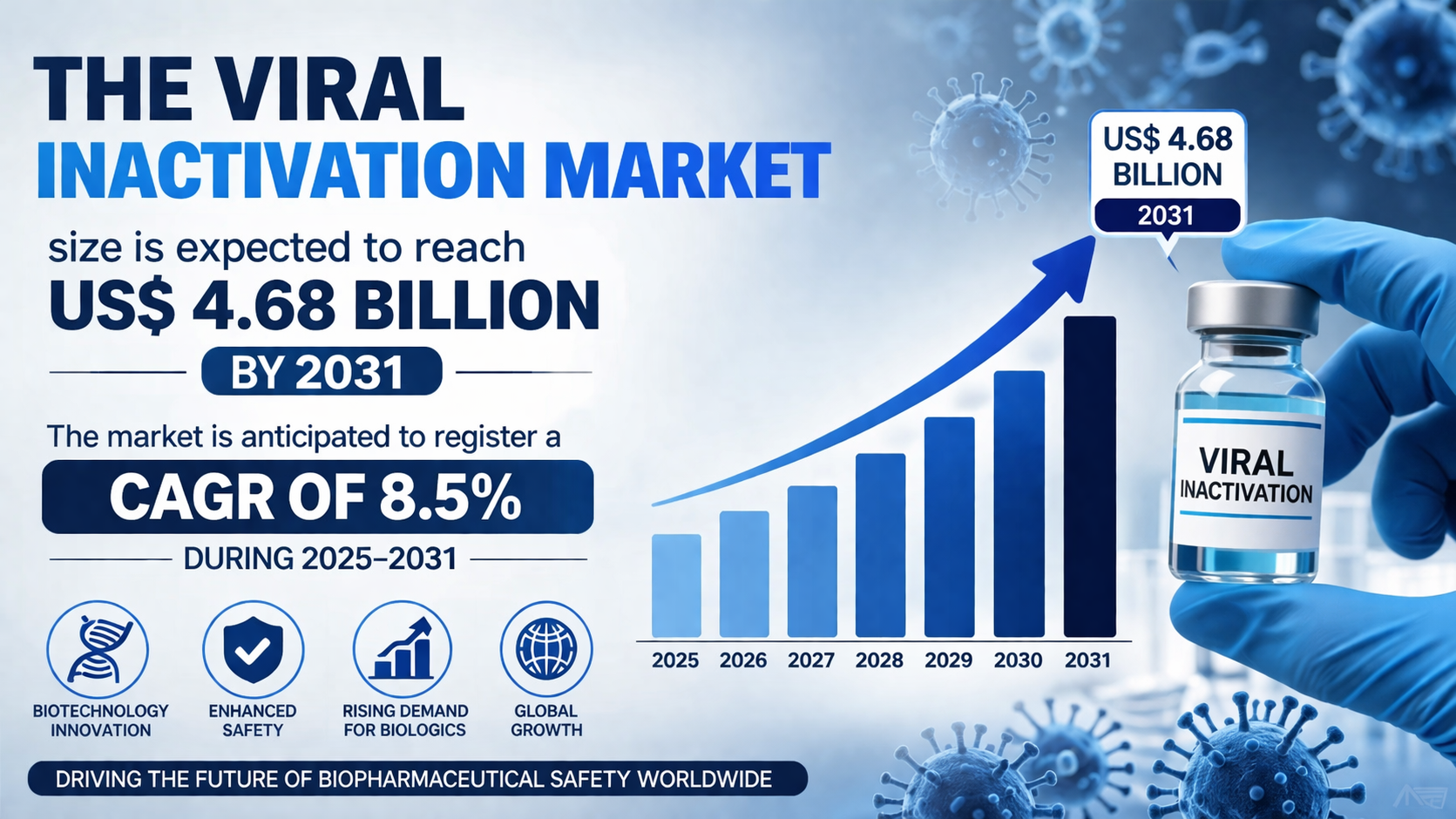
The Viral Inactivation Market Growth is gaining momentum due to expanding biopharmaceutical production and advancements in inactivation technologies. The Viral Inactivation Market size is expected to reach US$ 4.68 billion by 2031. The market is anticipated to register a CAGR of 8.5% during 2025–2031. This growth is driven by increasing demand for biologics and vaccines, along with the implementation of stringent regulatory frameworks. The focus on improving manufacturing efficiency and minimizing contamination risks is further contributing to market expansion.
Technological Advancements Driving Efficiency
Technological innovation is playing a pivotal role in shaping the Viral Inactivation Market. Methods such as solvent/detergent treatment, low pH treatment, ultraviolet (UV) irradiation, and pasteurization are widely used for effective viral elimination. Recent advancements in nanofiltration and membrane technologies are enhancing the efficiency of viral removal while preserving product integrity. Automation and digital monitoring systems are also improving process control and reducing human errors, making viral inactivation more reliable and efficient.
Download Sample PDF: https://www.theinsightpartners.com/sample/TIPRE00004176
Rising Demand for Vaccines and Biologics
The increasing global demand for vaccines and biologics is a major factor driving the Viral Inactivation Market. Immunization programs and the development of new vaccines for emerging infectious diseases have heightened the need for robust viral safety measures. Additionally, the growing use of monoclonal antibodies, gene therapies, and cell-based therapies is further boosting the demand for effective viral inactivation processes in biopharmaceutical manufacturing.
Growth of Outsourcing and Contract Services
The expansion of contract research organizations (CROs) and contract manufacturing organizations (CMOs) is contributing significantly to the growth of the Viral Inactivation Market. These organizations provide specialized viral inactivation services, enabling pharmaceutical companies to optimize production processes and reduce operational costs. The increasing trend of outsourcing in the pharmaceutical industry is expected to further drive demand for viral inactivation technologies and services.
Challenges and Market Constraints
Despite its strong growth prospects, the Viral Inactivation Market faces several challenges. High costs associated with advanced inactivation technologies and complex manufacturing processes can limit adoption, particularly among small and mid-sized companies. Additionally, maintaining the balance between effective viral elimination and preserving the quality of biologics remains a critical challenge. Stringent regulatory requirements also add complexity to the implementation of viral inactivation processes. However, ongoing advancements in technology are expected to address these challenges over time.
Key Players in the Viral Inactivation Market
-
Merck KGaA
-
Sartorius AG
-
Thermo Fisher Scientific Inc.
-
Danaher Corporation
-
Charles River Laboratories International, Inc.
-
Lonza Group AG
-
Parker Hannifin Corporation
-
Asahi Kasei Medical Co., Ltd.
-
Clean Cells SAS
-
Rad Source Technologies, Inc.
Future Outlook and Strategic Developments
The Viral Inactivation Market is expected to witness substantial growth in the coming years, driven by increasing demand for safe and effective biopharmaceutical products. The integration of advanced technologies such as artificial intelligence and automation is anticipated to enhance process efficiency and reliability. Strategic collaborations, mergers, and acquisitions among key players are likely to accelerate innovation and expand global reach. As regulatory standards continue to evolve and the demand for biologics rises, the market is poised for sustained expansion.
Related Reports
Automatic Pill Dispenser Market
Pharmaceutical Intermediates Market
About The Insight Partners
The Insight Partners delivers market intelligence and consulting services to help clients make informed decisions. The firm covers industries such as Aerospace and Defense, Automotive and Transportation, Semiconductor and Electronics, Biotechnology, Healthcare IT, Manufacturing, Medical Devices, Technology, Media, and Chemicals and Materials.
Contact Us
Email: sales@theinsightpartners.com
Website: www.theinsightpartners.com
Phone: +1-646-491-9876