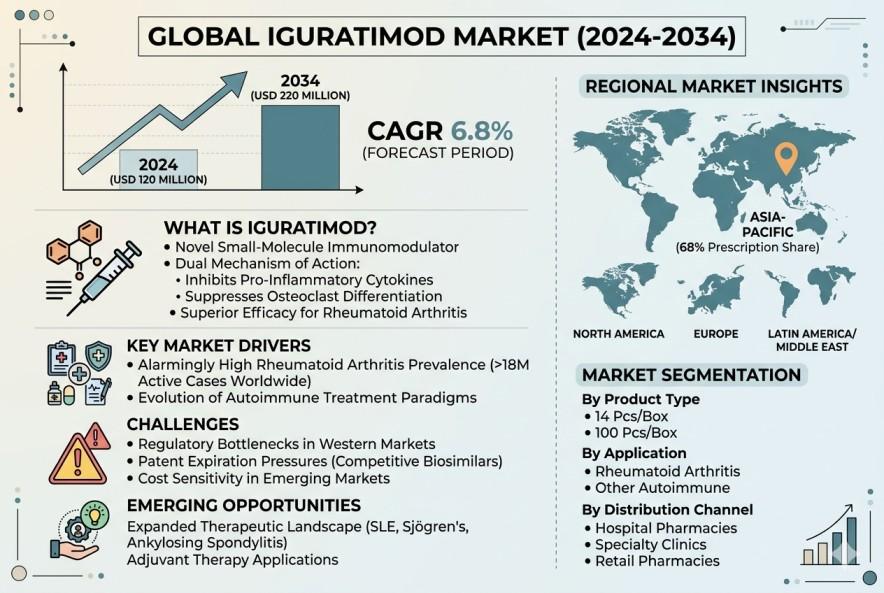
According to a new report from Intel Market Research, the global Iguratimod market was valued at USD 120 million in 2024 and is projected to reach USD 220 million by 2034, growing at a steady CAGR of 6.8% during the forecast period (2026-2034). This expansion reflects the drug's growing acceptance as a novel therapeutic option for autoimmune conditions, particularly rheumatoid arthritis, across major healthcare markets.
What is Iguratimod?
Iguratimod represents a breakthrough in small-molecule immunomodulation, specifically engineered to target the inflammatory pathways underlying autoimmune disorders. Unlike conventional disease-modifying antirheumatic drugs (DMARDs), it operates through a dual mechanism—simultaneously inhibiting pro-inflammatory cytokine production and suppressing osteoclast differentiation. This unique pharmacological profile has demonstrated superior efficacy in clinical settings, particularly for moderate-to-severe rheumatoid arthritis cases resistant to first-line therapies.
The drug's ability to reduce disease activity scores by 40-50% in clinical trials has positioned it as a valuable addition to rheumatologists' therapeutic arsenals. Its relatively favorable safety profile compared to biologics makes it particularly attractive for long-term disease management in aging populations.
📥 Download FREE Sample Report: Iguratimod Market - View in Detailed Research Report
Key Market Drivers
1. Alarmingly High Rheumatoid Arthritis Prevalence
The global burden of rheumatoid arthritis continues to escalate, with current estimates suggesting over 18 million active cases worldwide. This chronic condition's progressive nature creates sustained demand for effective treatment alternatives. Clinical studies have established Iguratimod's capacity to significantly improve joint function and reduce inflammation markers, making it particularly valuable for patients with inadequate responses to conventional DMARDs.
2. Evolution of Autoimmune Treatment Paradigms
Healthcare providers increasingly recognize the limitations of traditional immunomodulators, driving demand for targeted therapies with improved safety profiles. Iguratimod's novel mechanism—simultaneously addressing both inflammatory mediators and bone erosion processes—offers rheumatologists a sophisticated tool for comprehensive disease management. Market research indicates the Asia-Pacific region alone accounts for 68% of Iguratimod prescriptions, reflecting strong physician confidence in this advanced therapeutic approach.
Market Challenges
-
Regulatory Bottlenecks in Western Markets - Despite proven efficacy in Asian populations, Iguratimod faces significant approval hurdles in North America and Europe, where regulatory agencies require additional clinical data meeting their stringent evaluation criteria.
-
Patent Expiration Pressures - With key patents expiring in major markets, manufacturers face impending competition from biosimilars that could erode profit margins by 25-30% within the next five years.
-
Cost Sensitivity in Emerging Markets - The premium pricing strategy (20-35% higher than traditional DMARDs) limits accessibility in price-sensitive regions with underdeveloped reimbursement systems.
Emerging Opportunities
The therapeutic landscape for Iguratimod continues to expand beyond its core rheumatoid arthritis indication. Early-phase clinical trials demonstrate promising results in:
-
Systemic Lupus Erythematosus (SLE) - Showing 42% improvement in symptom management scores
-
Other Connective Tissue Disorders - Including Sjögren's syndrome and ankylosing spondylitis
-
Adjuvant Therapy Applications - Enhancing efficacy when combined with biological agents
These developments could unlock an additional $1.2 billion market potential by 2030, with research institutions and pharmaceutical companies actively investigating new therapeutic combinations and delivery mechanisms.
Regional Market Insights
-
Asia-Pacific: Commands dominant market share (68% of global prescriptions), led by Japan and China where regulatory approvals were secured earliest. The region benefits from established manufacturing infrastructure and high disease awareness.
-
North America: Shows accelerating adoption post-recent clinical validations, though market penetration remains constrained by reimbursement challenges and formulary restrictions.
-
Europe: Emerging as a high-growth market with several countries incorporating Iguratimod into step therapy protocols for treatment-resistant rheumatoid arthritis.
-
Latin America/Middle East: Demonstrates nascent but promising growth, driven by increasing rheumatology specialist density and improving healthcare access.
Market Segmentation
By Product Type
-
14 Pcs/Box (currently preferred format)
-
100 Pcs/Box
By Application
-
Rheumatoid Arthritis
-
Other Autoimmune Indications
By Distribution Channel
-
Hospital Pharmacies
-
Specialty Clinics
-
Retail Pharmacies
📘 Get Full Report Here: Iguratimod Market - View Detailed Research Report
Competitive Landscape
The market features a mix of established pharmaceutical leaders and innovative biotech firms, with three major players controlling approximately 75% of global Iguratimod sales. Strategic developments include:
-
Expansion of manufacturing capabilities in emerging markets
-
Development of next-generation formulations
-
Strategic licensing agreements to expand geographic reach
Key Companies Profiled:
-
Simcere Pharmaceutical
-
Eisai Co., Ltd.
-
Toyama Chemical
-
CSPC Pharmaceutical Group
-
Jiangsu Hengrui Medicine
Report Coverage Highlights
-
Comprehensive market size estimations and forecasts (2026-2034)
-
Detailed analysis of therapeutic application trends
-
Regulatory landscape and approval pathway analysis
-
Competitive intelligence and market share assessments
-
Emerging technology and formulation developments
📥 Download FREE Sample Report: Iguratimod Market - View in Detailed Research Report
About Intel Market Research
Intel Market Research is a leading provider of strategic intelligence, offering actionable insights in biotechnology, pharmaceuticals, and healthcare infrastructure. Our research capabilities include:
-
Real-time competitive benchmarking
-
Global clinical trial pipeline monitoring
-
Country-specific regulatory and pricing analysis
-
Over 500+ healthcare reports annually
Trusted by Fortune 500 companies, our insights empower decision-makers to drive innovation with confidence.
🌐 Website: https://www.intelmarketresearch.com
📞 Asia-Pacific: +91 9169164321
🔗 LinkedIn: Follow Us