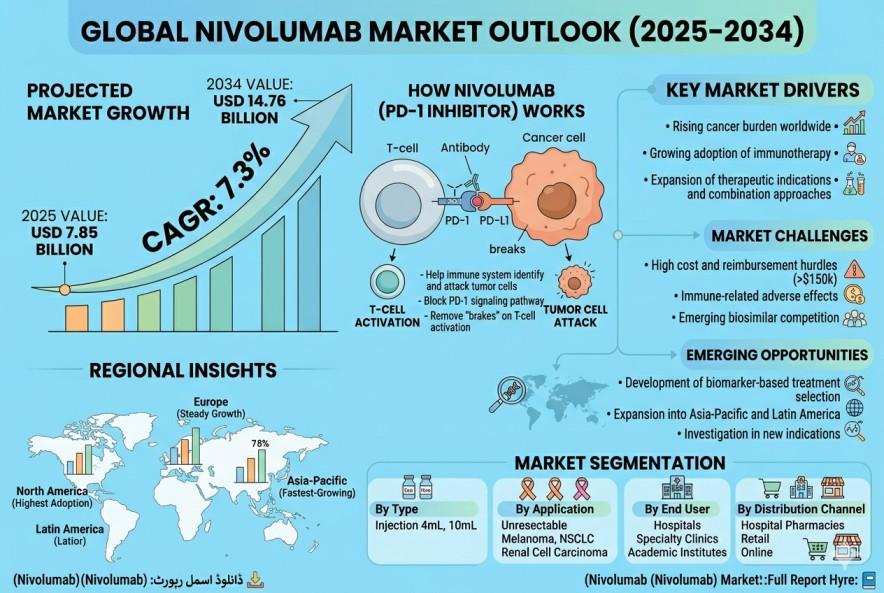
According to a new report from Intel Market Research, the global Nivolumab market was valued at USD 7.85 billion in 2025 and is projected to reach USD 14.76 billion by 2034, growing at a CAGR of 7.3% during the forecast period (2026-2034). This growth is driven by the increasing prevalence of cancer worldwide, expanding applications of immunotherapy, and growing adoption of PD-1 inhibitors in oncology treatment protocols.
What is Nivolumab?
Nivolumab is a groundbreaking PD-1 inhibitor immunotherapy that has revolutionized cancer treatment by helping the immune system recognize and attack tumor cells. As a fully human IgG4 monoclonal antibody, it specifically blocks PD-1 receptor signaling, effectively removing the "brakes" on T-cell activation. Currently approved for multiple indications including melanoma, NSCLC, and renal cell carcinoma, Nivolumab has demonstrated significant improvements in overall survival rates across several cancer types.
This comprehensive analysis provides complete insights into the global Nivolumab market, covering everything from macroeconomic trends to granular market details. The report examines market dynamics, competitive strategies, development patterns, niche segments, critical success factors, challenges, and comprehensive value chain analysis.
For industry participants, investors, and strategists, the report serves as an essential tool for understanding market positioning and identifying growth opportunities. It includes detailed competitive mapping of the Nivolumab landscape, featuring market shares, product performance metrics, and operational strategies of key players.
📥 Download Sample Report: Nivolumab Market - View in Detailed Research Report
Key Market Drivers
1. Rising Global Cancer Burden and Immunotherapy Adoption
The alarming increase in cancer incidence, with over 19 million new cases reported annually worldwide, has been a primary catalyst for Nivolumab's market expansion. Clinical studies consistently demonstrate its ability to improve survival rates by 30-40% across multiple cancer types. A landmark study published in the Journal of Clinical Oncology (2020) showed Nivolumab nearly doubled 5-year survival rates in advanced melanoma patients compared to traditional therapies.
2. Broadening Treatment Indications and Combination Approaches
Regulatory approvals continue to expand Nivolumab's therapeutic scope. Recent FDA clearances for hepatocellular carcinoma and adjuvant melanoma treatment have significantly increased eligible patient populations. Combination therapies, particularly with ipilimumab, show synergistic effects that enhance treatment efficacy:
-
Melanoma - Combination therapy demonstrates superior progression-free survival in advanced cases
-
Renal Cell Carcinoma - Shows improved objective response rates versus monotherapy
-
NSCLC - Emerging data suggests benefits in specific biomarker-positive populations
These developments underscore Nivolumab's versatility as both standalone and combination therapy in modern oncology practice.
Market Challenges
-
High Cost and Reimbursement Hurdles - With annual treatment costs often exceeding $150,000, access remains limited in price-sensitive markets. Many healthcare systems struggle to justify the expenditure despite demonstrated clinical benefits.
-
Immune-Related Adverse Effects - The drug's immune-mediated toxicities (occurring in 15-20% of patients) require specialized management protocols that increase overall treatment costs and complexity.
-
Emerging Biosimilar Competition - Impending patent expirations and biosimilar development could significantly alter market dynamics by 2025-2026.
Emerging Opportunities
The immunotherapy landscape presents multiple growth avenues for Nivolumab, particularly in developing regions and novel therapeutic areas. Key opportunities include:
-
Expansion into Asian and Latin American markets where cancer incidence is rising faster than treatment access
-
Development of biomarker-based treatment selection to improve cost-effectiveness
-
Investigation in 15+ new indications including rare cancers through ongoing clinical trials
-
Innovations in drug delivery systems and administration protocols
These factors collectively create a favorable environment for market expansion and technological innovation in Nivolumab applications.
📥 Download Sample PDF: Nivolumab Market - View in Detailed Research Report
Regional Market Insights
-
North America: Dominates the global market with the highest adoption rates, driven by favorable reimbursement policies and advanced healthcare infrastructure. The US accounts for the majority of regional sales.
-
Europe: Shows steady growth with Germany and UK leading in clinical innovation. EMA approvals continue to expand treatment access across EU markets.
-
Asia-Pacific: Emerging as the fastest-growing region with Japan and China making significant investments in immunotherapy infrastructure.
-
Latin America: Brazil and Argentina show promising adoption trends as healthcare systems gradually incorporate advanced oncology therapies.
-
Middle East & Africa: GCC countries demonstrate higher adoption rates, while Sub-Saharan Africa remains largely untapped due to economic constraints.
Market Segmentation
By Type
-
Injection 4mL
-
Injection 10mL
By Application
-
Unresectable Melanoma
-
Metastatic Melanoma
-
Metastatic Squamous NSCLC
-
Classical Hodgkin Lymphoma
-
Renal Cell Carcinoma
By End User
-
Hospitals
-
Specialty Clinics
-
Academic Research Institutes
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
📘 Get Full Report Here: Nivolumab Market - View Detailed Research Report
Competitive Landscape
The Nivolumab market is dominated by Bristol-Myers Squibb and Ono Pharmaceutical through their flagship product Opdivo®. However, the competitive environment is evolving with several pharmaceutical giants advancing their immunotherapy pipelines.
The report provides detailed competitive analysis of key players including:
-
Bristol-Myers Squibb
-
Ono Pharmaceutical
-
Merck & Co.
-
Roche Holding AG
-
Novartis AG
-
Emerging biosimilar developers
Report Deliverables
-
Comprehensive market forecasts from 2026 to 2034
-
In-depth analysis of pipeline developments and regulatory trends
-
Competitive intelligence and market share analysis
-
Pricing dynamics and reimbursement scenario analysis
-
Detailed segmentation by product type, application, and geography
📘 Get Full Report Here: Nivolumab Market - View Detailed Research Report
About Intel Market Research
Intel Market Research is a leading provider of strategic intelligence, offering actionable insights in biotechnology, pharmaceuticals, and healthcare infrastructure. Our research capabilities include:
-
Real-time competitive benchmarking
-
Global clinical trial pipeline monitoring
-
Country-specific regulatory and pricing analysis
-
Over 500+ healthcare reports annually
Trusted by Fortune 500 companies, our insights empower decision-makers to drive innovation with confidence.
🌐 Website: https://www.intelmarketresearch.com
📞 Asia-Pacific: +91 9169164321
🔗 LinkedIn: Follow Us